I am Professor of Membrane Protein Structural Biology and joined the Biosciences Institute at Newcastle University in January 2013. Before this, I was a faculty member within the Program in Molecular Medicine at UMass Medical School in Worcester, Massachusetts (USA). The main focus of my research is to understand cell-surface processes in Gram-negative bacteria, and in particular how small molecules are transported across the outer membrane (OM). For this, we determine the three-dimensional structures of bacterial OM proteins by X-ray crystallography and cryo-EM. Those structures are then used, in combination with functional data obtained from biochemical experiments, to propose transport models that can be tested experimentally. We also collaborate with computational biologists and mass spectrometrists to gain additional insights in small-molecule transport. Since becoming a PI in 2004, my laboratory has solved the crystal structures of more than 100 OM proteins, via the timeline shown below.
The location of the OM is shown by the grey bars. A short description of the organism and transport are given on the slides. PDB codes are given as well.
1. Understanding small-molecule uptake by the Bacteroidetes.
The Bacteroidetes are the most abundant Gram-negative phylum of our microbiota and therefore play a key role in shaping the bacterial communities in our bodies. The aim of this project, funded by the Wellcome Trust, is to understand how small molecules are taken up across the outer membrane (OM) in members of the Bacteroidetes via active transport and passive diffusion. As the first step in the utilisation of any small molecule nutrient, OM translocation is a critical determinant of microbial fitness in the competitive environment of the gut. The outcomes of this project will complement “big data” studies to hopefully allow rational manipulation of the human gut microbiota to advance human health.
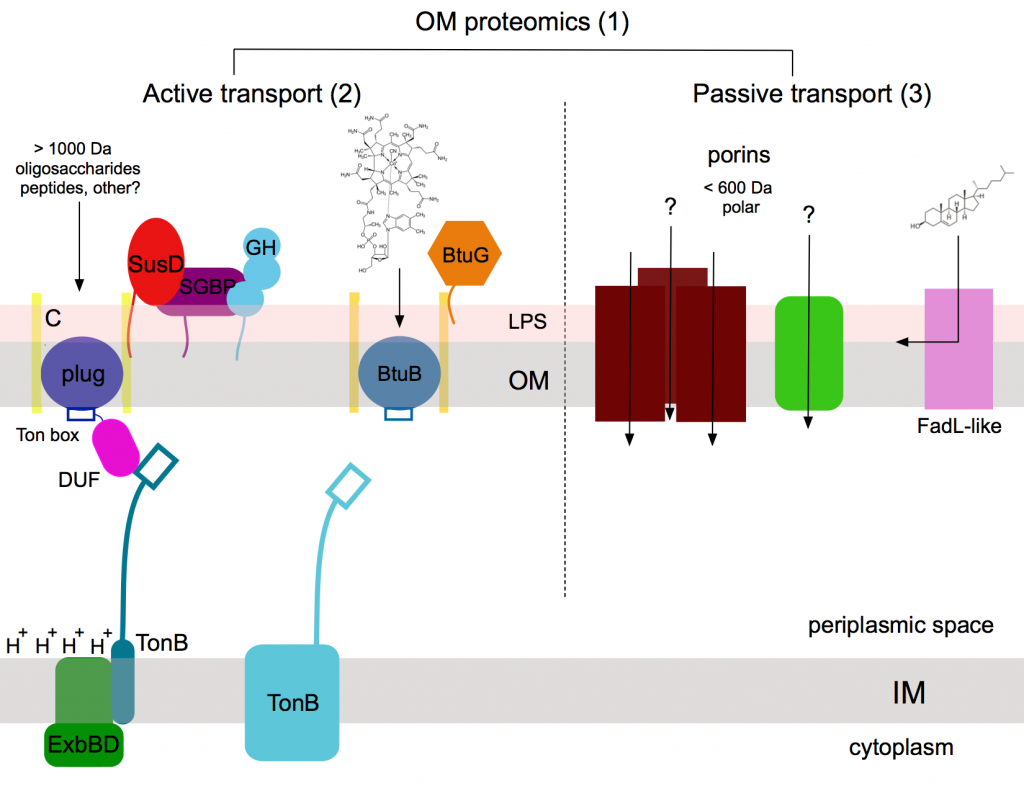
Figure 1 Schematic overview of OM transport in Bacteroides thetaiotaomicron. Areas of interest not only include glycan and oligopeptide uptake by SusCD systems and vitamin B12 uptake by BtuBG complexes, but also how various TonB-ExbBD complexes in the IM provide the energy for OM active transport. We also study the passive transport of small molecules, with an emphasis on FadL-like channels that may mediate uptake of fatty acids, bile acids and sterols. An overarching goal of the project will be to gain insight in protein abundance under various conditions via quantitative OM proteomics.
Movie 1 Dynamics of the RagAB transporter from Porphyromonas gingivalis. Cryo-EM map of open-closed (OC) RagAB, with cartoon models shown for RagA (blue) and RagB (yellow or grey) subunits. The cryo-EM structures provide direct evidence for a “pedal bin” transport mechanism, which also operates for glycan uptake by SusCD transporters.
Selected publications:
- Glenwright AJ et al. (2017) Structural basis for nutrient acquisition by dominant members of the human gut microbiota. Nature 541, 407-411.
- Bolam DN, van den Berg B (2018) TonB-dependent transport by the gut microbiota: novel aspects of an old problem. Curr Opin Struct Biol. 51, 35-43.
- Madej M et al. (2020) Structural and functional insights into oligopeptide acquisition by the RagAB transporter from Porphyromonas gingivalis. Nat Microbiol. doi: 10.1038/s41564-020-0716-y.
- Because FadL orthologs are found in many biodegrading bacteria, our research is relevant for the bioremediation of xenobiotics within the environment.
2. Maintenance of OM lipid asymmetry
The increasing emergence of pathogenic (Gram-negative) bacteria that are resistant towards antibiotics represents a big future threat for public health, a situation that has recently been likened to a “ticking time bomb” and a possible “apocalypse” by the chief medical officer of England The maintenance of lipid asymmetry (Mla) system, which is present in most Gram-negative bacteria, ensures that the permeability barrier of the OM is maintained by removing phospholipids from the outer leaflet. The MlaA protein is the OM component of the Mla system. We have determined the first structures of MlaA-porin complexes, which, together with functional data, provides mechanistic insights. MlaA forms a donut in the inner leaflet of the outer membrane, which binds phospholipids (PL) from the outer leaflet and removes these via a central channel, somewhat similar to a vacuum cleaner. Our current work, funded by the BBSRC, focuses on determining the PL translocation mechanism through MlaA and how PL are transferred to the periplasmic protein MlaC.
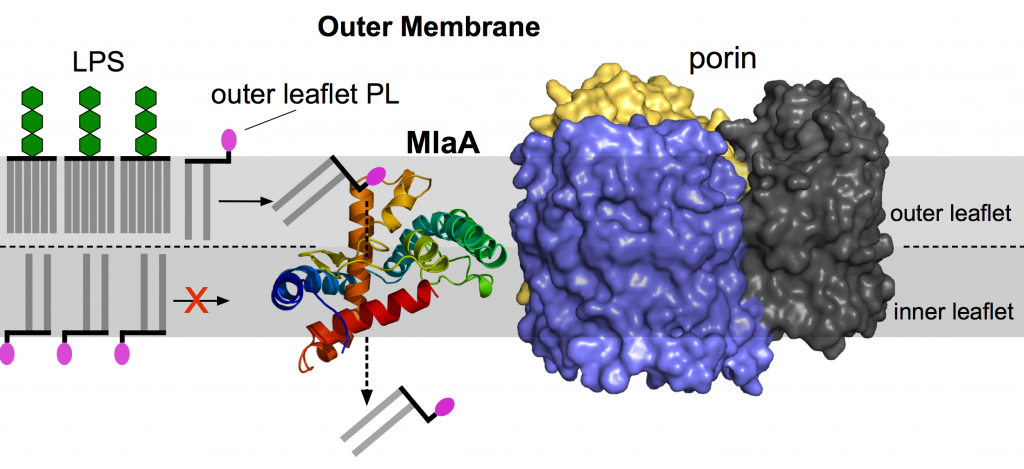
Figure 2 Model for PL removal from the OM outer leaflet by MlaA.
Selected publication:
- Abellón-Ruiz J et al. (2017) Structural basis for maintenance of bacterial outer membrane lipid asymmetry. Nat Microbiol. 2, 1616-1623.
3. Transport of hydrophobic molecules
A long-standing interest of the lab is how hydrophobic molecules are taken up across the OM by members of the FadL channel family. Due to the presence of lipopolysaccharide (LPS) on the cell surface, the OM is a very efficient barrier for hydrophobic molecules, in contrast to regular phospholipid bilayers. Using the archetype of the family (E. coli FadL) as a model system, we have shown that uptake of long-chain fatty acids (LCFA) occurs via a unique mechanism that we have termed lateral diffusion. Here, the substrates diffuse laterally into the OM via an opening in the channel wall. Very recently, we have shown that uptake of monoaromatic hydrocarbons (MAH) such as toluene by biodegrading bacteria also occurs via lateral diffusion. Future work will focus on the role of FadL channels in quorum sensing and gut biology.
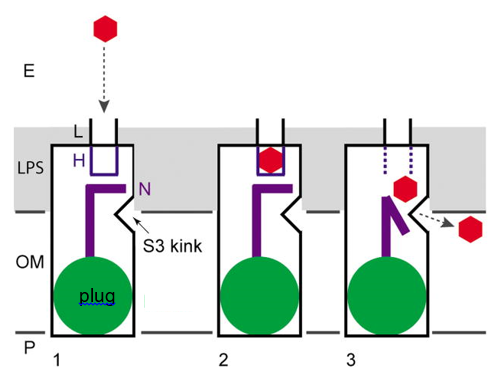
Figure 3 Schematic model of the lateral diffusion mechanism for FadL channels. The hydrophobic substrate (red hexagon) moves into the OM via the lateral opening in the barrel wall. In this way, the polar barrier of the LPS layer (grey) is bypassed. The plug domain (green) inside the barrel prevents direct diffusion into the periplasmic space.
Movie 2 Lateral diffusion pathway for benzene through TodX from Pseudomonas putida F1.
Selected publications:
- van den Berg B et al. (2004) Crystal structure of the long-chain fatty acid transporter FadL. Science 304, 1506-9.
- Hearn EM et al. (2009) Transmembrane passage of hydrophobic compounds through a protein channel wall. Nature 458, 367-70.
- Somboon S et al. (2020) Uptake of monoaromatic hydrocarbons during biodegradation by FadL channel-mediated lateral diffusion. https://www.biorxiv.org/content/10.1101/2020.05.11.088534v1.full
Collaborators:
- Prof. Carol Robinson, University of Oxford
- Prof. Andy Goodman, Yale
- Prof. Neil Ranson, University of Leeds
- Prof. Jan Potempa, Jagiellonian University
- Prof. Syma Khalid, University of Southampton
- Prof. Ulrich Kleinekathöfer, Jacobs University Bremen