As a Structural Biologist, my main research interest is in the determination and understanding of the structure of proteins associated to human disease, particularly those caused by infectious agents. Our research focuses on understanding the molecular details of interactions between pathogenic and human host cells that are crucial for infections. Of particular interest are surface proteins from Candida albicans and Clostridium difficile, both important health concerns as they are responsible for emerging hospital acquired infections.
Clostridium difficile S-layer
Clostridium difficile is a leading cause of hospital acquired infections worldwide, causing considerable economic pressure on healthcare systems. C. difficile strains are notoriously resistant to antibiotics, and there are currently no vaccines against infection. Recent outbreaks have seen the emergence of strains that cause even more severe disease and exhibit a higher mortality rate. These gram-positive bacteria have an outside para-crystalline layer that is presumed to act as a protective shield and that is thought to be implicated in virulence.
In collaboration with Prof Neil Fairweather and Dr Robert Fagan, we focus on understanding S-layer organisation, primarily by determining the structure of the major S-layer protein constituent, SlpA.
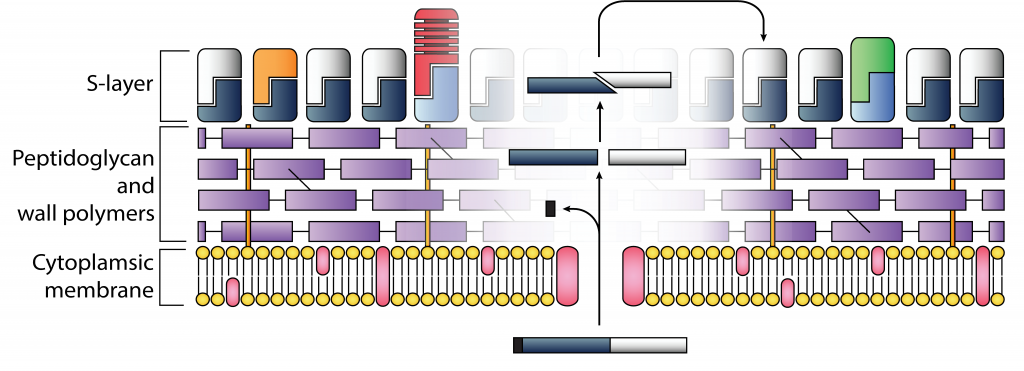

We are also studying other cell wall proteins identified in C. difficile, aiming to understand their macromolecular details with our structural studies, therefore complementing the biological characterisation carried out by our collaborators.
As part of our general interest in pathogen cell surface proteins, we have recently established collaborations to study C. difficile spore surface proteins. Of particular interest are proteins involved in spore-mother cell interaction as well as key surface enzymes.
Collaborators: Prof Neil Fairweather (Imperial College London), Dr Robert Fagan (Sheffield University), Prof Adriano O. Henriques (ITQB, Portugal)
Fungal biofilms
Biofilms are matrix-enclosed microbial masses that adhere to biological or non-biological surfaces and have been implicated in both the spread and onset of infections. Importantly, they provide a protective environment as microorganisms within the biofilm are more resistant to antibiotics and host defences.
Candida albicans can cause a wide range of diseases and the formation of biofilms on catheters and medical implants is a significant source of systemic infections in the hospital environment.
The CFEM-containing family of proteins, characterised by an eight-cysteine motif unique to fungal species, has been implicated in biofilm formation and could provide a structural scaffold for new rational drug development. Our research, in collaboration with Dr Carol Munro at the University of Aberdeen, is focusing on providing details into the structural organisation of this unusual motif and understand its role in candidal infections.
Collaborators: Dr Carol Munro (University of Aberdeen)
Recently, the structure of the first candidal adhesion, Als9 was determined, which allowed the identification of a flexible unusual binding cleft that accounted for the observed relatively unspecific ligand binding nature of these adhesins.
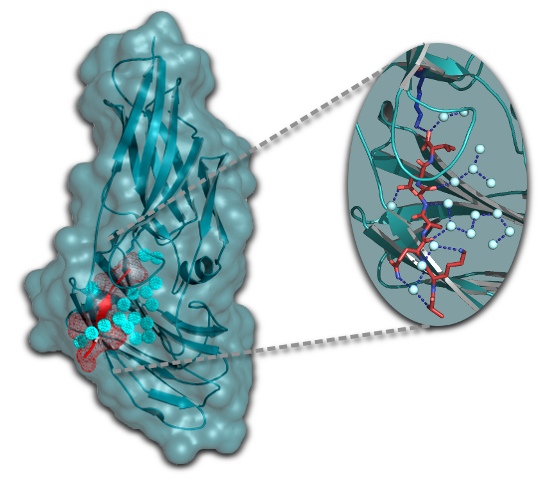
Salgado, P.S., Yan, R., Taylor, J.D., Buchnell, L., Jones, R., Hoyer, L., Matthews, S.J., Simpson, P., Cota, E. (2011) Structural basis for the broad specificity to host-cell ligands by the pathogenic fungus Candida albicans. Proc Natl Acad Sci USA 108, 15775-9. [Pubmed]
Biofilm characterisation
As part of our ongoing interest in biofilm formation and characterisation, we have recently been awarded funding for an interdisciplinary pilot project to further characterise Candida albicans ability to adhere to various surfaces, such as dentures, catheters and surgical instruments. Based on previous work on the key C. albicans protein family Als (see above) we aim to use advanced AFM and XPS techniques to quantify the strength of binding between the C. albicans protein and a range of surfaces (polymeric/inorganic). By better understanding the nature of the binding of this protein family to different surfaces, we hope to identify material/surface properties responsible for promoting strong binding and those which prevent binding. This should then allow the rational design materials to which candidal cells can not adhere.
Collaborators: Dr Mark Birch (ICM), Dr Jinju Chen (MSE), Dr David Fulton (Chem), Dr Matt German (Dentistry), Prof Sudpita Roy (CEAM) and Dr Ernest Cota (Imperial College London).